Opinion statement
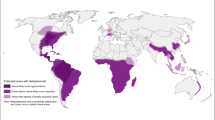
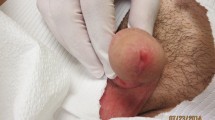
Leishmaniasis, a protozoal infection transmitted by sandfly bite, produces a clinical spectrum of disease ranging from asymptomatic infection to ulcerative skin and mucosal lesions to visceral involvement. Leishmaniasis is endemic in regions of Africa, the Middle East, south Asia, southern Europe, northern South America, and Central America. There has been an increase in imported leishmaniasis into developed, non-endemic countries due to increasing global travel. While pentavalent antimonials have been the mainstay of antileishmanial treatment for decades, newer therapeutic options have become available for all forms of infection, including liposomal amphotericin B, miltefosine, fluconazole, and ketoconazole. For the returning traveler with cutaneous leishmaniasis in the USA, treatment approaches are determined based on infecting species, initial presentation, extent and progression of disease, the advantages and drawbacks of available parenteral and oral drugs, and clinician-consultant experience.




Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Pavli A, Maltezou HC. Leishmaniasis, an emerging infection in travelers. Int J Infect Dis. 2010;14(12):e1032–9.
Rodrigues JC, Godinho JL, de Souza W. Biology of human pathogenic trypanosomatids: epidemiology, lifecycle and ultrastructure. Subcell Biochem. 2014;74:1–42.
Goto H, Lauletta Lindoso JA. Cutaneous and mucocutaneous leishmaniasis. Infect Dis Clin North Am. 2012;26(2):293–307.
Reithinger R, Dujardin JC, Louzir H, Pirmez C, Alexander B, Brooker S. Cutaneous leishmaniasis. Lancet Infect Dis. 2007;7(9):581–96.
Murray HW, Berman JD, Davies CR, Saravia NG. Advances in leishmaniasis. Lancet. 2005;366(9496):1561–77.
Murray HW. Leishmaniasis in the United States: treatment in 2012. Am J Trop Med Hyg. 2012;86(3):434–40. This paper reviews the new treatment options available to U.S. clinicians who are treating patients who contracted leishmaniasis abroad.
Solomon M, Benenson S, Baum S, Schwartz E. Tropical skin infections among Israeli travelers. Am J Trop Med Hyg. 2011;85(5):868–72.
Jirmanus L, Glesby MJ, Guimarães LH, Lago E, Rosa ME, Machado PR, et al. Epidemiological and clinical changes in American integumentary leishmaniasis in an area of Leishmania (Viannia) braziliensis transmission over a 20-year period. Am J Trop Med Hyg. 2012;86(3):426–33.
Shirian S, Oryan A, Hatam GR, Panahi S, Daneshbod Y. Comparison of conventional, molecular, and immunohistochemical methods in diagnosis of typical and atypical cutaneous leishmaniasis. Arch Pathol Lab Med. 2014;138(2):235–40.
McCall LI, Zhang WW, Ranasinghe S, Matlashewski G. Leishmanization revisited: immunization with a naturally attenuated cutaneous Leishmania donovani isolate from Sri Lanka protects against visceral leishmaniasis. Vaccine. 2013;31(10):1420–5.
Blum J, Buffet P, Visser L, Harms G, Bailey MS, Caumes E, et al. LeishMan recommendations for treatment of cutaneous and mucosal leishmaniasis in travelers, 2014. J Travel Med. 2014;21(2):116–29.
Hodiamont CJ, Kager PA, Bart A, de Vries HJ, van Thiel PP, Leenstra T, et al. Species-directed therapy for leishmaniasis in returning travellers: a comprehensive guide. PLoS Negl Trop Dis. 2014;8(5):e2832.
Morizot G, Kendjo E, Mouri O, Thellier M, Pérignon A, Foulet F, et al. Travelers with cutaneous leishmaniasis cured without systemic therapy. Clin Infect Dis. 2013;57(3):370–80. This study by the Cutaneous Leishmaniasis French Study Group reviewed clinical outcomes of various treatment paradigms for CL and described clinical criteria for starting systemic versus topical/local therapy.
Neves LO, Talhari AC, Gadelha EP, Silva Júnior RM, Guerra JA, Ferreira LC, et al. A randomized clinical trial comparing meglumine antimoniate, pentamidine and amphotericin B for the treatment of cutaneous leishmaniasis by Leishmania guyanensis. An Bras Dermatol. 2011;86(6):1092–101. This study is one of the largest randomized control studies in recent years to compare clinical outcomes in parenteral therapy for NWCL.
Mueller Y, Nguimfack A, Cavailler P, Couffignal S, Rwakimari JB, Loutan L, et al. Safety and effectiveness of amphotericin B deoxycholate for the treatment of visceral leishmaniasis in Uganda. Ann Trop Med Parasitol. 2008;102(1):11–9.
Ben Salah A, Ben Messaoud N, Guedri E, Zaatour A, Ben Alaya N, Bettaieb J, et al. Topical paromomycin with or without gentamicin for cutaneous leishmaniasis. N Engl J Med. 2013;368(6):524–32.
Khalili G, Dobakhti F, Mahmoudzadeh-Niknam H, Khaze V, Partovi F. Immunotherapy with Imiquimod increases the efficacy of glucantime therapy of Leishmania major infection. Iran J Immunol. 2011;8(1):45–51.
David CV, Craft N. Cutaneous and mucocutaneous leishmaniasis. Dermatol Ther. 2009;22(6):491–502.
Bumb RA, Satoskar AR. Radiofrequency-induced heat therapy as first-line treatment for cutaneous leishmaniasis. Expert Rev Anti Infect Ther. 2011;9(6):623–5.
Bumb RA, Prasad N, Khandelwal K, Aara N, Mehta RD, Ghiya BC, et al. Long-term efficacy of single-dose radiofrequency-induced heat therapy vs. intralesional antimonials for cutaneous leishmaniasis in India. Br J Dermatol. 2013;168(5):1114–9.
López L, Robayo M, Vargas M, Vélez ID. Thermotherapy an alternative for the treatment of American cutaneous leishmaniasis. Trials. 2012;13:58.
Arevalo J, Ramirez L, Adaui V, Zimic M, Tulliano G, Miranda-Verástegui C, et al. Influence of Leishmania (Viannia) species on the response to antimonial treatment in patients with American tegumentary leishmaniasis. J Infect Dis. 2007;195(12):1846–51.
Oliveira LF, Schubach AO, Martins MM, Passos SL, Oliveira RV, Marzochi MC, et al. Systematic review of the adverse effects of cutaneous leishmaniasis treatment in the New World. Acta Trop. 2011;118(2):87–96.
Monge-Maillo B, López-Vélez R. Therapeutic options for old world cutaneous leishmaniasis and new world cutaneous and mucocutaneous leishmaniasis. Drugs. 2013;73(17):1889–920.
Wortmann G, Zapor M, Ressner R, Fraser S, Hartzell J, Pierson J, et al. Lipsosomal amphotericin B for treatment of cutaneous leishmaniasis. Am J Trop Med Hyg. 2010;83(5):1028–33. This review of patients treated at the Walter Reed Army Medical Center demonstrated high efficacy (84%) of liposomal amphotericin B against various species of Leishmania.
Solomon M, Pavlotzky F, Barzilai A, Schwartz E. Liposomal amphotericin B in comparison to sodium stibogluconate for Leishmania braziliensis cutaneous leishmaniasis in travelers. J Am Acad Dermatol. 2013;68(2):284–9.
Schuster FL, Guglielmo BJ, Visvesvara GS. In-vitro activity of miltefosine and voriconazole on clinical isolates of free-living amebas: Balamuthia mandrillaris, Acanthamoeba spp., and Naegleria fowleri. J Eukaryot Microbiol. 2006;53(2):121–6.
Machado PR, Penna G. Miltefosine and cutaneous leishmaniasis. Curr Opin Infect Dis. 2012;25(2):141–4.
Rubiano LC, Miranda MC, Muvdi Arenas S, Montero LM, Rodríguez-Barraquer I, Garcerant D, et al. Noninferiority of miltefosine versus meglumine antimoniate for cutaneous leishmaniasis in children. J Infect Dis. 2012;205(4):684–92. This paper presents data from a recent randomized noninferiority study comparing oral miltefosine to antimony in the treatment of NWCL. Based on this and other trials in Colombia and Brazil, miltefosine was approved by the FDA for the indication of CL, ML, and VL.
Dorlo TP, van Thiel PP, Schoone GJ, Stienstra Y, van Vugt M, Beijnen JH, et al. Dynamics of parasite clearance in cutaneous leishmaniasis patients treated with miltefosine. PLoS Negl Trop Dis. 2011;5(12):e1436.
Emad M, Hayati F, Fallahzadeh MK, Namazi MR. Superior efficacy of oral fluconazole 400 mg daily versus oral fluconazole 200 mg daily in the treatment of cutaneous leishmania major infection: a randomized clinical trial. J Am Acad Dermatol. 2011;64(3):606–8. This paper presents data from a randomized trial in Iran which evaluated the efficacy of high-dose fluconazole for the treatment of L. major OWCL, and demonstrated an 81% rate of complete resolution after 6 weeks of treatment.
Sousa AQ, Frutuoso MS, Moraes EA, Pearson RD, Pompeu MM. High-dose oral fluconazole therapy effective for cutaneous leishmaniasis due to Leishmania (Vianna) braziliensis. Clin Infect Dis. 2011;53(7):693–5.
Saenz RE, Paz H, Berman JD. Efficacy of ketoconazole against Leishmania braziliensis panamensis cutaneous leishmaniasis. Am J Med. 1990;89(2):147–55.
Ramanathan R, Talaat KR, Fedorko DP, Mahanty S, Nash TE. A species-specific approach to the use of non-antimony treatments for cutaneous leishmaniasis. Am J Trop Med Hyg. 2011;84(1):109–17. This case series from the National Institutes of Health evaluated efficacy and tolerability of non-antimony-based treatments for CL, including oral miltefosine, ketoconazole, and liposomal amphotericin B.
Compliance with Ethics Guidelines
Conflict of Interest
Daniel P. Eiras declares that he has no conflict of interest.
Laura A. Kirkman declares that she has no conflict of interest.
Henry W. Murray declares that he has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Travel Medicine
Rights and permissions
About this article
Cite this article
Eiras, D.P., Kirkman, L.A. & Murray, H.W. Cutaneous Leishmaniasis: Current Treatment Practices in the USA for Returning Travelers. Curr Treat Options Infect Dis 7, 52–62 (2015). https://doi.org/10.1007/s40506-015-0038-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-015-0038-4